The Structure of Metal
- By Rick Groleau
- Posted 04.30.02
- NOVA
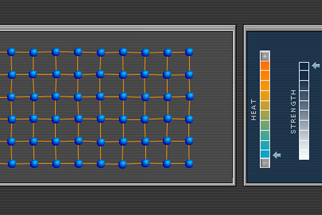
From carrying current on a computer's circuit boards to holding up skyscrapers, metal has countless uses, thanks to its special characteristics: It's hard and strong, yet it's bendable. It can be welded to other pieces of metal and rolled flat or hammered into shape. It conducts heat and electricity. It has a lustrous surface. At the atomic heart of metal is a crystalline structure—tightly packed atoms arranged in orderly rows. This feature explores this heart and reveals what it is that gives metal its special characteristics and how metal behaves under the stress of heat and outside forces.
This feature originally appeared on the site for the NOVA program Building on Ground Zero.
Sources
Materials Science and Engineering: An Introduction, by William D. Callister, Jr. John Wiley & Sons, 1994
"The Nature of Metals," by A. H. Cottrell. Scientific American, September, 1967
The Science of Structures and Materials, by J. E. Gordon. Scientific American Library, 1988
Ceramics, Plastics, and Metals: An Introduction to the Science of Solids, by Richard H. Krock and Merrill L. Ebner. D.C. Heath & Co., 1965.
Credits
Images
- (all)
- © NOVA/WGBH Educational Foundation
Related Links
-

Making Stuff: Stronger
David Pogue tests his mettle against the world's strongest stuff, from steel and Kevlar to bioengineered silk.
-

Materials That Changed History
From ceramics to steel, paper to plastics, certain basic substances have long propped up civilization.
-

The World's Strongest Stuff
From diamonds to spider silk, see some of the hardest, strongest, and toughest materials on Earth.
-

Twin Towers of Innovation
A host of engineering marvels distinguished the original World Trade Center.
You need the Flash Player plug-in to view this content.